General Information about Sulfur
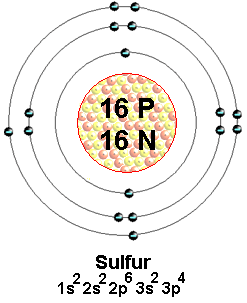
- 1. Element name and symbol: Sulfur, S
- 2. Atomic Number: 16
- 3. Average Atomic Mass: 32.07 amu
- 4. Number of protons, neutrons, and electrons: 16, 16, 16
5. Location on periodic table (Group, Period, Block): 3, 16, P 6. Chemical family: Nonmetal
7. Electron Configuration: 1s^2 2s^2 2p^6 3s^2 3p^4
8. Atomic radius: 103 pm
9. Ionization energy: 999.6 kJ/mol
10. Electronegativity value: 2.58 Paulings
11. Color, texture, and physical state at room temperature: solid yellow microcrystals
12. Boiling point and melting point: 444.6 °C and 115.2 °C
13. Valence electrons: 6
14. Common ions formed: +2/-2
